440 Trastuzumab For Injection
Price 17999 INR/ Box
440 Trastuzumab For Injection Specification
- Indication
- Metastatic Breast Cancer, Gastric Cancer
- Salt Composition
- Trastuzumab Lyophilized Powder
- Dosage Form
- For Injection
- Enzyme Types
- Monoclonal Antibody (not enzyme)
- Feature
- Highly purified monoclonal antibody targeting HER2 protein
- Ingredients
- Trastuzumab, Excipients
- Application
- Intravenous infusion for cancer treatment
- Ph Level
- Neutral pH (typically 6.07.0 in reconstituted solution)
- Physical Color/Texture
- White to off-white lyophilized powder
- Fermentation Smell
- Odorless
- Enzymatic Activity
- Not applicable (monoclonal antibody)
- Storage Instructions
- Store at 28C, protect from light, do not freeze
- Shelf Life
- 24 months
- Reconstitution Solvent
- Sterile Water for Injection
- Packing Type
- Single vial with carton packaging
- Container Material
- Medical Grade Glass
- Marketed By
- Licensed Oncology Pharmaceuticals Company
- Intended Patient Group
- Adults with confirmed HER2-positive tumors
- Clinical Trials
- Backed by large scale multicenter studies
- Registration
- CDSCO/WHO-GMP approved
- Product Code
- TRZ440
- Compatibility
- Do not mix with other drugs in same IV line
- Expiry Details
- Marked on label and carton
- Usage Precaution
- For hospital or clinical use only, under physician supervision
- Administration Route
- Intravenous
- Batch Number
- Available on carton
- Strength
- 440 mg per vial
About 440 Trastuzumab For Injection
Premier Monoclonal Antibody Therapy with Distinct Advantages
440 Trastuzumab For Injection stands out as a first-class choice for HER2-positive cancer therapy, backed by large-scale multicenter clinical trials. Its neutral pH and highly purified monoclonal antibody ensure safety and efficacy. Competitive advantages include CDSCO/WHO-GMP certification and advanced, medical-grade glass packaging. Application areas extend across major oncology centers, clinical research institutes, and specialized hospitals, making it perfect for handling both metastatic breast and gastric cancer cases with confidence.
Certified Sample Access and Smooth Goods Transport Experience
Experience the premium service of sample availability for 440 Trastuzumab, ensuring that clinical settings can verify quality before bulk procurement. Shipped goods are monitored for temperature control, ensuring preservation of potency and stability throughout transport. All products are dispatched from certified and licensed suppliers, delivering international-standard pharmaceuticals with relevant certifications such as CDSCO and WHO-GMP. Flexible payment terms are available, making transactions straightforward and securing your hospital's access to world-class cancer therapy.
FAQ's of 440 Trastuzumab For Injection:
Q: How should 440 Trastuzumab For Injection be administered for optimal results?
A: This medication is designed for intravenous infusion, following reconstitution with sterile water for injection. Administration should only be performed by healthcare professionals in a hospital or clinical setting under strict physician supervision.Q: What clinical benefits does this product offer for cancer patients?
A: 440 Trastuzumab targets the HER2 protein on cancer cells, helping to inhibit tumor growth specifically for HER2-positive metastatic breast and gastric cancer patients. It is supported by extensive multicenter clinical trials, ensuring proven effectiveness and safety.Q: When is it appropriate to consider 440 Trastuzumab For Injection in therapy?
A: The product is intended for use in adults diagnosed with confirmed HER2-positive metastatic breast or gastric cancer, as part of a comprehensive cancer treatment plan decided by an oncologist.Q: Where can healthcare providers source this first-class oncology product?
A: Licensed hospitals, clinics, and authorized distributors in India and globally can obtain 440 Trastuzumab For Injection from registered suppliers and exporters with proper certifications and established reputation.Q: What precautions must be taken during preparation and administration?
A: Do not mix 440 Trastuzumab with other drugs in the same IV line, and always reconstitute with sterile water. Store the reconstituted vial at 2-8C, protect it from light, and never freeze. Usage is restricted to clinical environments.Q: How can buyers access product sample and certification details?
A: Samples can be requested from verified distributors. All shipped goods come with complete batch information, expiry, and CDSCO/WHO-GMP certification documents, ensuring compliance and quality assurance.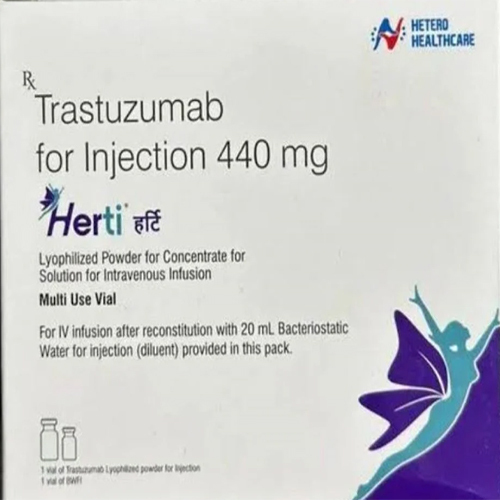

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Injection Category
500 Mg - 50 Ml Rituximab
Price 6500 INR / Vial
Minimum Order Quantity : 1 Vial
Shelf Life : 24 months
Salt Composition : Rituximab 500 mg
Indication : NonHodgkins lymphoma, Chronic lymphocytic leukemia, Rheumatoid arthritis, Granulomatosis with polyangiitis, Microscopic polyangiitis
Dosage Form : Injection
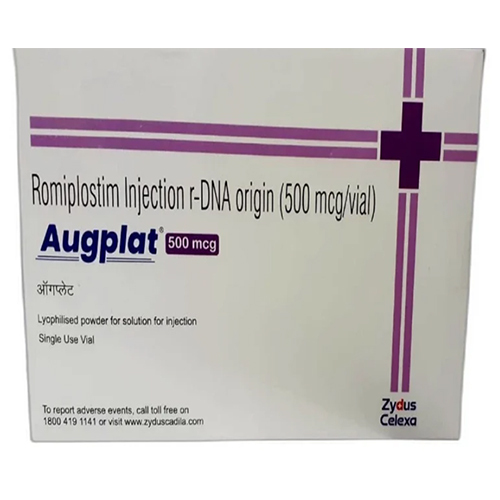
500 Mcg Romiplostim Injection R-DNA Origin
Price 4500 INR / Vial
Minimum Order Quantity : 10 Vials
Shelf Life : 24 months
Salt Composition : Romiplostim 500 Mcg
Indication : Treatment of chronic immune thrombocytopenia (ITP)
Dosage Form : Injection
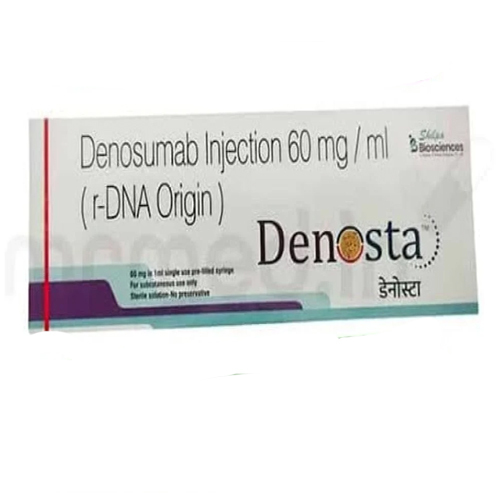
60 Mg Denosumab Injection
Price 6500 INR / Piece
Minimum Order Quantity : 1 Piece
Shelf Life : 24 months
Salt Composition : Denosumab 60 mg
Indication : Treatment of osteoporosis, bone loss due to hormone therapy, and prevention of skeletalrelated events in patients with bone metastases
Dosage Form : Injection
50 Mg Doxorubicin Hydrochloride IP
Price 49 INR / Vial
Minimum Order Quantity : 1 Vial
Shelf Life : 24 months from manufacturing date.
Salt Composition : Doxorubicin Hydrochloride IP
Indication : Anticancer agent used in the treatment of various malignancies including leukemia, lymphoma, and solid tumors.
Dosage Form : Injection
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese