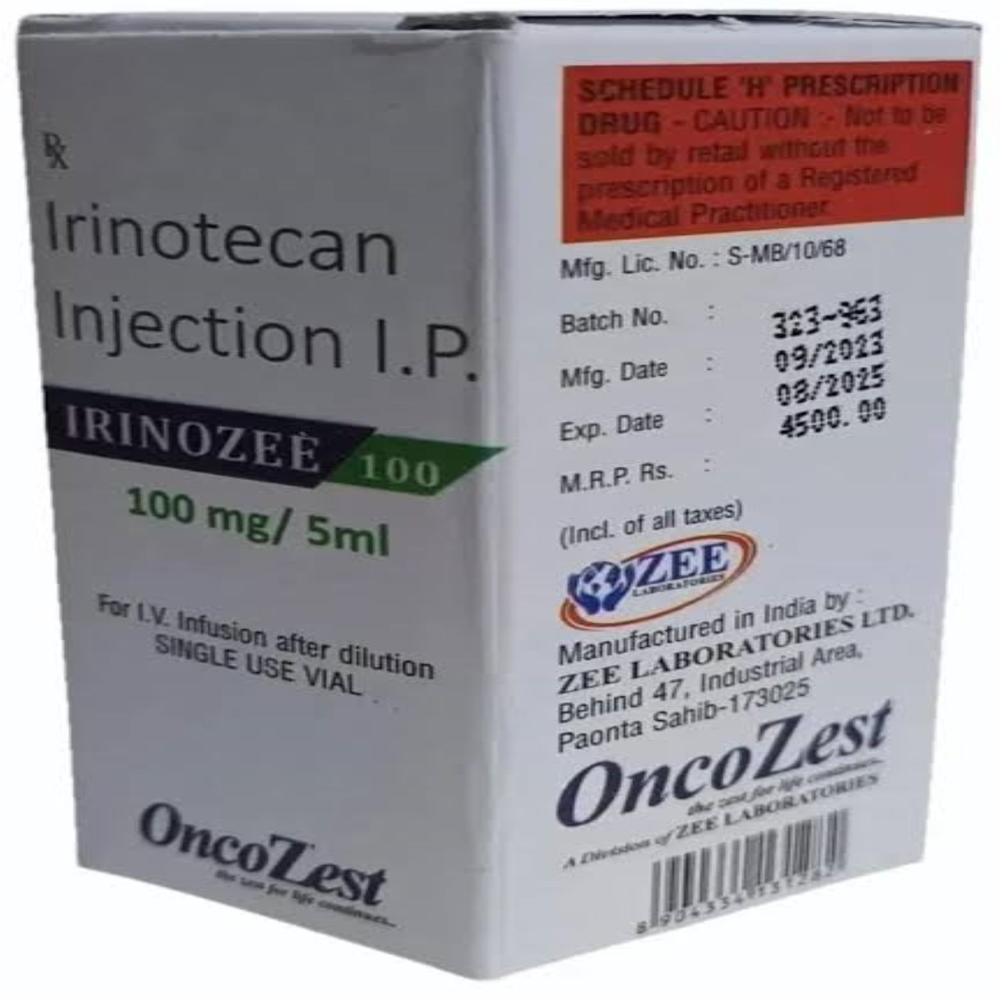
irinotecan 40 mg
Price 120.0 INR/ Vial
irinotecan 40 mg Specification
- Dosage Form
- Injection
- Salt Composition
- Irinotecan Hydrochloride Trihydrate
- Indication
- Used in the treatment of colorectal cancer
- Enzyme Types
- Topoisomerase I inhibitor
- Feature
- Sterile, preservative-free, ready-to-use
- Ingredients
- Irinotecan Hydrochloride Trihydrate, excipients
- Application
- For intravenous use only, under medical supervision
- Ph Level
- Approximately 3.5
- Physical Color/Texture
- Clear, pale yellow solution
- Fermentation Smell
- Odorless
- Enzymatic Activity
- Not an enzyme
- Storage Instructions
- Store below 25C. Protect from light. Do not freeze.
- Shelf Life
- 24 months from manufacture
- Prescription/OTC Status
- Prescription only
- Incompatibilities
- Do not mix with solutions containing glucose
- Container Volume
- 2 mL per vial
- USP/EP Compliance
- Complies with relevant pharmacopeia
- Precautions
- Use with caution in hepatic impairment and pregnancy
- Packaging Type
- Glass vial with rubber stopper and flip-off cap
- Strength
- 40 mg per 2 mL vial
- ATC Code
- L01XX19
- Method of Administration
- IV infusion after dilution
- Primary Use
- Advanced or metastatic colorectal carcinoma
- Diluent
- Dilute with 0.9% Sodium Chloride or 5% Dextrose
About irinotecan 40 mg
Key Advantages and Clinical Applications of Irinotecan 40 mg
Irinotecan 40 mg offers distinct advantages, including compliance with international pharmacopeia and a ready-to-use formulation. Its primary application is in intravenous treatment under medical supervision for colorectal cancer, especially in advanced or metastatic stages. The product stands out for its pivotal role in comprehensive cancer therapy, consistent potency, and convenience of use in hospital, oncology clinics, and institutional settings. Its enhanced safety profile and sterile packaging further augment its competitive edge for healthcare professionals.
Supply Ability and Market Reach for Irinotecan 40 mg
This product is distributed, exported, and traded both domestically and across export markets. With a robust supply ability tailored to meet urgent oncology requirements, vials are shipped and delivered on promptly scheduled timelines. Orders are efficiently processed, ensuring quick handover to partners nationwide as well as priority export handoffs. Reliable supply chains and strategic distribution guarantee wide market coverage within India and beyond, fulfilling the needs of the pivotal healthcare sector.
FAQ's of irinotecan 40 mg:
Q: How should Irinotecan 40 mg be prepared and administered?
A: Irinotecan 40 mg should be diluted with 0.9% sodium chloride or 5% dextrose before intravenous infusion. It must not be mixed with glucose-containing solutions and should only be administered under medical supervision.Q: What are the key prescription requirements for Irinotecan 40 mg?
A: This medication is available by prescription only and should be prescribed by an oncologist or qualified healthcare professional, ensuring proper medical oversight and dosage adherence.Q: When is Irinotecan 40 mg used in the clinical setting?
A: Irinotecan 40 mg is primarily indicated for the treatment of advanced or metastatic colorectal carcinoma, usually within hospital, oncology center, or clinical settings.Q: Where can Irinotecan 40 mg be stored and for how long?
A: Store the vials below 25C, protected from light. Do not freeze. The shelf life is 24 months from the date of manufacture.Q: What are the main competitive benefits of Irinotecan 40 mg?
A: This product offers USP/EP compliance, a sterile preservative-free formula, ready-to-use convenience, and reliable efficacy, making it a preferred choice for clinicians treating colorectal cancer.Q: How quickly can Irinotecan 40 mg be shipped to domestic and export markets?
A: With efficient logistics, shipments are typically processed and handed over promptly for both domestic and export markets, ensuring timely delivery to hospitals and clinics.


Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Medicines Category
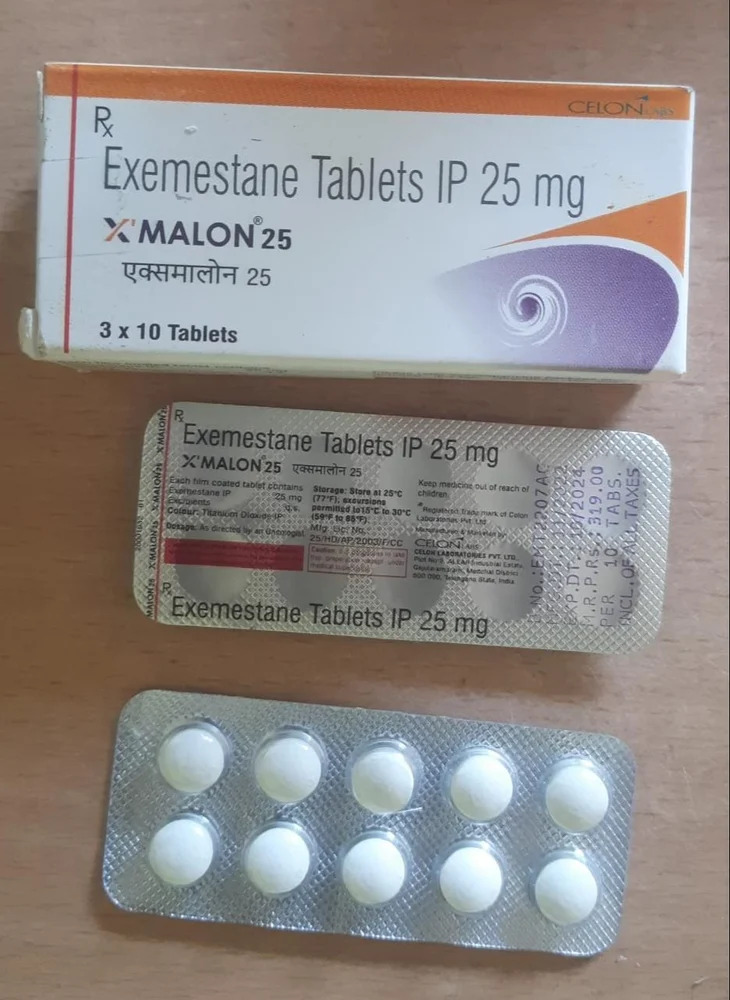
25mg Exemestane Tablets IP
Price 250 INR / Unit
Minimum Order Quantity : 10 Units
Storage Instructions : Dry place
Shelf Life : Up to 24 Months
Salt Composition : Exemestane
Origin : India
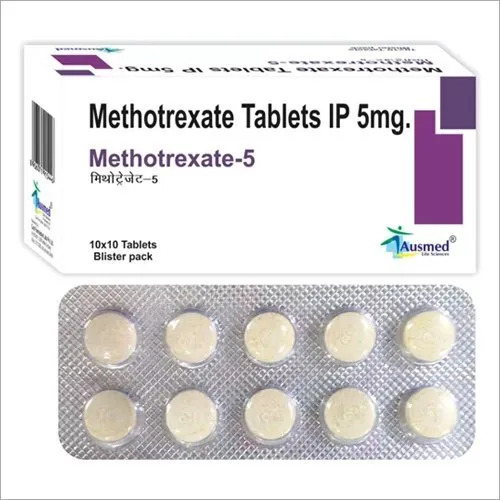
50mg MethotreXate Tablets IP
Price 25 INR / Strip
Minimum Order Quantity : 5 Strips
Storage Instructions : Dry place
Shelf Life : Up to 24 Months
Salt Composition : Methotrexate
Origin : India
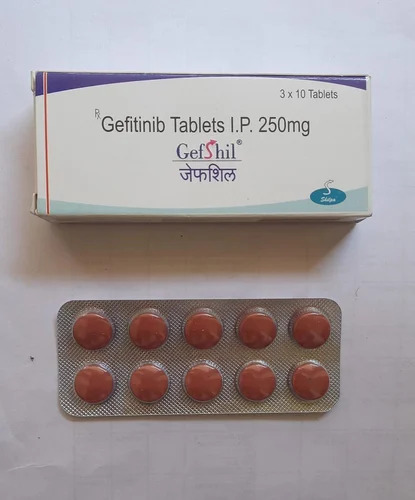
250mg Gefitinib Tablets IP
Price 500 INR / Strip
Minimum Order Quantity : 10 Packs
Storage Instructions : Dry place
Shelf Life : Up to 24 Months
Salt Composition : Gefitinib
Origin : India
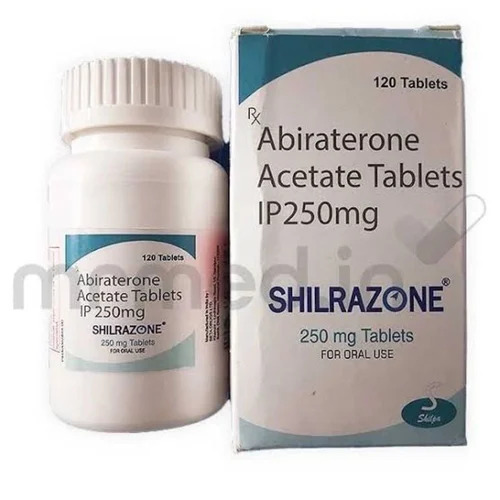
250mg Abiraterone Acetate Tablets
Price 5000 INR / Pack
Minimum Order Quantity : 1 Pack
Storage Instructions : Dry place
Shelf Life : Up to 24 Months
Salt Composition : Abiriton Acetate
Origin : India
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese