Docetaxel Injection IP
Price 300 INR/ Vial
Docetaxel Injection IP Specification
- Dosage Form
- Injection
- Salt Composition
- Docetaxel
- Indication
- Breast cancer, non-small cell lung cancer, prostate cancer, gastric cancer, head and neck cancer
- Feature
- Sterile, single-use, cytotoxic injectable
- Ingredients
- Docetaxel, polysorbate 80, ethanol anhydrous, water for injection
- Application
- Ph Level
- Neutral (pH 3.05.0)
- Physical Color/Texture
- Clear, colorless to pale yellow solution
- Fermentation Smell
- Characteristic, alcoholic smell
- Storage Instructions
- Store below 25C, protect from light
- Shelf Life
- 24 months
- Regulatory Classification
- Schedule H (India)
- Precautions
- Use under physician supervision, cytotoxic precautions required
- Packaging Type
- Glass vial with flip-off seal
- Compatibility
- Use only with recommended diluents and IV sets
- Prescription Status
- Prescription only (Rx)
- Route of Administration
- Intravenous
- Solubility
- Soluble in polysorbate 80 and ethanol
- Pack Size
- Single-dose vial
- Available Strengths
- 20 mg/0.5 ml, 80 mg/2 ml, 120 mg/3 ml
About Docetaxel Injection IP
Directions, Usage, and Indications of Docetaxel Injection IP
Docetaxel Injection IP is intended for intravenous administration only and must be used strictly as directed by a qualified healthcare provider. This prescription-only medicine serves as a potent chemotherapeutic agent for treating breast, non-small cell lung, prostate, gastric, and head and neck cancers. It is designed for general hospital and clinical use under cytotoxic precautions, ensuring patient safety and efficacy. Adherence to specified diluents and administration sets is essential for optimal therapeutic outcomes.
Export Market, Delivery, and Packaging Details for Docetaxel Injection IP
Docetaxel Injection IP is available for domestic distribution across India and holds a robust presence in the export market. Packaging includes a sterile, glass vial with a flip-off seal to guarantee product integrity during delivery and storage. Each order is promptly dispatched following strict sample policy and regulatory compliance, ensuring timely and safe supply to distributors and hospitals. Shelf life is guaranteed for 24 months when stored below 25C and protected from light.
FAQ's of Docetaxel Injection IP:
Q: How should Docetaxel Injection IP be administered safely?
A: Docetaxel Injection IP is to be administered intravenously by a trained healthcare professional in a clinical or hospital setting. Strict adherence to cytotoxic safety precautions and the use of recommended diluents and IV sets is required.Q: What are the main indications for using Docetaxel Injection IP?
A: This medication is primarily indicated for the treatment of cancers such as breast, non-small cell lung, prostate, gastric, and head and neck cancers.Q: When should Docetaxel Injection IP be used?
A: Docetaxel Injection IP should be used strictly under a physician's supervision and only after being prescribed as part of a comprehensive anti-cancer chemotherapy protocol.Q: Where can I purchase or distribute Docetaxel Injection IP?
A: Docetaxel Injection IP is available through licensed distributors, exporters, suppliers, and traders across India, complying with Schedule H regulations.Q: What precautions must be taken during the handling and storage of Docetaxel Injection IP?
A: Handle with cytotoxic precautions, store below 25C, protect from light, and always use sterile techniques to maintain product efficacy and safety.Q: What are the benefits of choosing Docetaxel Injection IP for chemotherapy?
A: It offers first-class, reliable, and effective treatment options for various cancers, packaged in single-dose sterile vials that ensure both safety and convenience.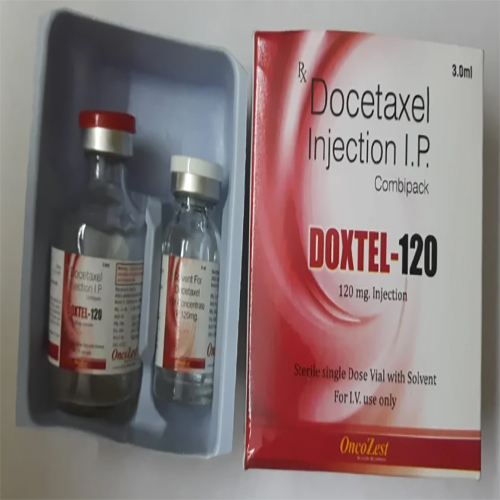

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Injection Category
Filgrastim Injection IP
Price 249 INR / Piece
Minimum Order Quantity : 1 Piece
Storage Instructions : Store between 2C 8C; protect from light, do not freeze
Indication : Neutropenia, to promote neutrophil production in patients undergoing chemotherapy, bone marrow transplant, or HIV infection.
Salt Composition : Filgrastim
Shelf Life : 24 months
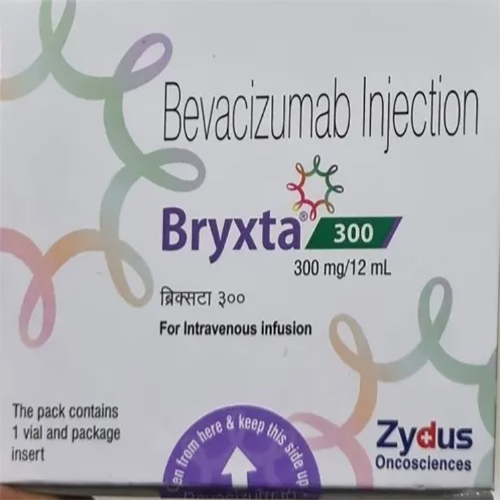
300 Mg - 12 Ml Bevacizumab Injection
Price 10000 INR / Vial
Minimum Order Quantity : 1 Vial
Storage Instructions : Store refrigerated at 2C to 8C. Do not freeze.
Indication : Cancer Treatment (Colorectal Cancer, Lung Cancer, Renal Cell Carcinoma, Glioblastoma)
Salt Composition : Bevacizumab (300mg) per 12ml vial
Shelf Life : 24 months from manufacturing date
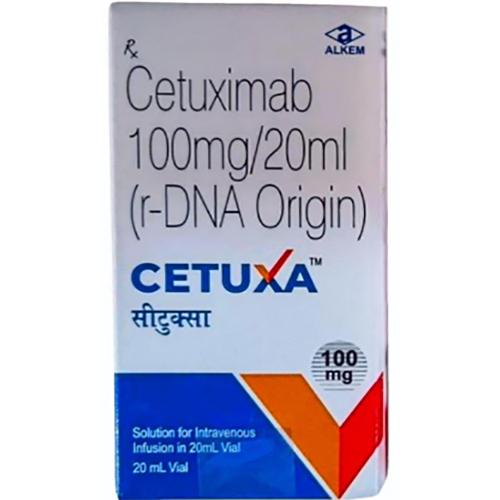
100 Mg Cetuximab Injection
Price 7000 INR / Vial
Minimum Order Quantity : 1 Vial
Storage Instructions : Store at 2C to 8C, do not freeze
Indication : Colorectal cancer, Head and neck cancer
Salt Composition : Cetuximab 100mg
Shelf Life : 24 months
Azacitidine For Injection
Price 2000 INR / Vial
Minimum Order Quantity : 1 Vial
Storage Instructions : Store below 30C, protect from light and moisture
Indication : Treatment of Myelodysplastic Syndromes (MDS), Chronic Myelomonocytic Leukemia (CMML), and Acute Myeloid Leukemia (AML)
Salt Composition : Azacitidine
Shelf Life : 24 months from the date of manufacture
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese